Healthcare
Psilocybin’s Antidepressant Effects Can Last Over 12 Months Says New Medical Study
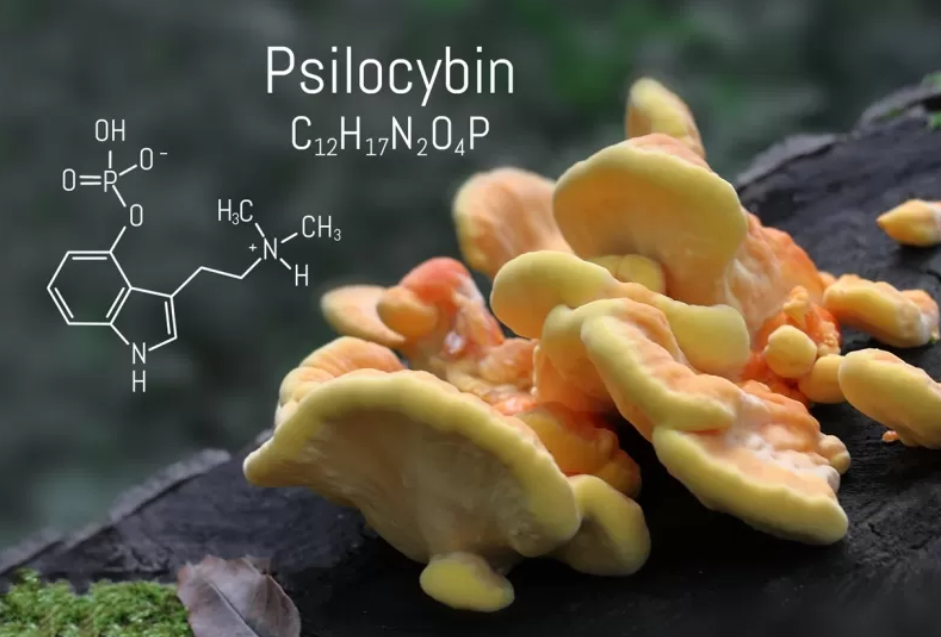
Psilocybin’s Antidepressant Effects Are Long Lasting, Says Studies
Millions of people around the world rely on antidepressant medications just to get by.
There are dozens of different antidepressants in the pharmaceutical market, and in order to feel its effects, many need to be taken daily for up to 2 weeks to feel any significant improvements. In addition, these pills can often take from 3 up to 6 months for one to no longer feel depressed, if they even work for you. There are countless people who are unable to rely on the effects of antidepressants because they simply don’t work for them, or make them feel numb; oftentimes, antidepressants can even make you feel worse than you did to start.
The side effects of antidepressants vary: these can include insomnia, fatigue, headaches, diarrhea or constipation, dry mouth, and sexual problems, to name a few. Since depression can truly rob you of quality of life – so either way, it’s a risk.
But what if you only had to take one thing ONCE or twice a year, and notice significant improvements in depression already?
Psilocybin mushrooms may be able to do just that.
A study from earlier this year involved 27 participants, all of whom had a long history with depression. Many were experiencing symptoms of depression for around 2 years before they were recruited for the study, conducted by researchers at Johns Hopkins University, who were following up on previous studies that revealed psilocybin treatment was effective in treating depressive symptoms for up to a month.
Among the participants, 88% were treated with conventional antidepressant medications in the past while 58% said they were using antidepressants currently. After they were all screened, they were divided into two groups randomly, made to attend preparatory meetings, then given two doses of psilocybin 2 weeks apart from August 2017 through April 2019. They also came back to the Johns Hopkins Bayview Medical Center for follow-ups a day and a week after all sessions, followed by follow ups at 1, 3, 6, and 12 month intervals after the 2nd session. Twenty-four of these participants were able to complete all the psilocybin and follow-up visits.
According to the researchers, psilocybin treatment among both groups revealed that there were significant reductions in depression symptoms. The symptoms were also found to stay low for not just one and 3 months, but up to 12 months following treatment.
“Our findings add to evidence that, under carefully controlled conditions, this is a promising therapeutic approach that can lead to significant and durable improvements in depression,” explains Natalie Gukasyan, MD, the Johns Hopkins University School of Medicine assistant professor of psychiatry and behavioral sciences. She does warn that “the results we see are in a research setting and require quit a lot of preparation and structured support from trained clinicians and therapists, and people should not attempt to try it on their own.”
“Psilocybin not only produces significant and immediate effects, it also has a long duration, which suggests that it may be a uniquely useful new treatment for depression,” adds Roland Griffiths, Ph.D., founding director of the Johns Hopkins Center for Psychedelic and Consciousness Research, and a Professor in Neuropsychopharmacology of Consciousness. “Compared to standard antidepressants, which must be taken for long stretches of time, psilocybin has the potential to enduringly relieve the symptoms of depression with one or two treatments.”
Given the track record of standard antidepressants, this is incredible news.
However, the researchers highlight the importance of doing more research to verify that psilocybin is indeed effective enough to last for more than 12 months.
Older Studies
There have been similar older studies done that show similar results.
For one, NYU Grossman School of Medicine researchers followed up on the results of their 2016 study which found that just a one-time treatment of single dose psilocybin was effective in delivering significant improvements for the mental well-being of patients who had cancer.
The cancer patients suffered from existential dread, anxiety, depression, and hopelessness. However, after following up 6.5 months after treatment, they found that psilocybin treatment was associated with better quality of life, improvement in spiritual wellbeing, and decreases in anxiety and depression.
“Our results represent the strongest evidence to date of a clinical benefit from psilocybin therapy, with the potential to transform care for patients with cancer-related psychological distress,” explains Stephen Ross, MD, the study’s lead author. “If larger clinical trials prove successful, then we could ultimately have available a safe, effective, and inexpensive medication – dispensed under strict control – to alleviate the distress that increases suicide rates among cancer patients,” Ross adds.
One of the most important findings of their study was that the clinical scores for depression and anxiety lasted for 8 months – which was far longer than the monitoring period for the study. Study co-author Anthony Bossis, PhD, also said that the patients reported greater improvements in quality of life following psilocybin treatment: they had more energy, went out more, performed better at work, and had better relationships with family members. Some patients even said that they were more spiritual in various ways, experienced unusual peacefulness, and had feelings of altruism.
“Our study showed that psilocybin facilitated experiences that drove reductions in psychological distress,” Bossis said. “And if it’s true for cancer care, then it could apply to other stressful medical conditions.”
Conclusion
Psilocybin is proving to be a remarkable, revolutionary compound in the world of medicine. As of today, it already has found so many far-reaching uses with deep impacts on the lives of people struggling with emotional and mental health. It might not be a panacea, but it is a fantastic starting point that may potentially have long-lasting effects.
Education News
AIIMS Gorakhpur Reservation Controversy: FIR Alleges Fake OBC Certificate Used Despite ₹80 Lakh Income
A major controversy has emerged at AIIMS Gorakhpur after allegations surfaced that reservation benefits were misused to secure a postgraduate medical seat. A criminal case has been registered against former AIIMS Gorakhpur Executive Director Dr. G.K. Pal and his son, Dr. Oro Prakash Pal, over the alleged use of a forged Other Backward Class (OBC) Non-Creamy Layer certificate for admission to an MD course.
The case has triggered widespread debate within medical and administrative circles, raising serious questions about transparency and oversight in admissions to premier medical institutions.
FIR Filed on Court’s Direction
The First Information Report (FIR) was registered at the AIIMS police station following directions from Chief Judicial Magistrate Tvishi Srivastava. According to the complaint, the alleged offence took place on August 30, 2024, when forged documents were purportedly used to obtain an MD seat under the OBC reservation quota.
Sources indicate that the matter was subsequently brought to the attention of the Union Ministry of Health, prompting internal reviews and administrative action.
Alleged Income Far Above Eligibility Threshold
Central to the allegations is the claim that Dr. G.K. Pal and his wife Parvati Pal have a combined annual income exceeding ₹80 lakh. Under existing reservation rules, families with such income levels are not eligible for OBC Non-Creamy Layer benefits.
Following the emergence of the controversy, Dr. Pal was first removed from his position at AIIMS Gorakhpur and later relieved of responsibilities at AIIMS Patna. He is currently posted at JIPMER Puducherry. With the registration of the FIR, officials suggest that further legal and departmental action may follow, depending on the outcome of the investigation.
Complaint Highlights Systemic Concerns
The complaint was filed by Ashutosh Kumar Mishra, a resident of Divyanagar in the Cantonment area. He alleged that the events related to the case occurred between January and September 2024. The complainant argued that misuse of reservation provisions by individuals in senior positions undermines the integrity of public institutions and erodes trust in the medical education system.
The case has reignited concerns over whether verification mechanisms for reservation certificates are robust enough, particularly in high-stakes admissions.
Impact on Medical Community
The allegations have sent shockwaves through the medical fraternity. Experts believe that if the claims are substantiated, the case could prompt a broader review of admission procedures and lead to stricter scrutiny of category certificates across institutions like AIIMS.
Authorities have stated that all relevant documents will be carefully examined before taking further steps. The outcome of the investigation is expected to have far-reaching implications for accountability and compliance in medical admissions.
Questions Await Answers
As the probe continues, several key issues remain unresolved: whether reservation norms were deliberately violated, how verification processes failed, and whether similar cases may surface in the future. For now, the focus remains on the investigation and its potential consequences.
Business
New Mexico cannabis operator fined, loses license for alleged BioTrack fraud

New Mexico regulators fined a cannabis operator nearly $300,000 and revoked its license after the company allegedly created fake reports in the state’s traceability software.
The New Mexico Cannabis Control Division (CCD) accused marijuana manufacturer and retailer Golden Roots of 11 violations, according to Albuquerque Business First.
Golden Roots operates the The Cannabis Revolution Dispensary.
The majority of the violations are related to the Albuquerque company’s improper use of BioTrack, which has been New Mexico’s track-and-trace vendor since 2015.
The CCD alleges Golden Roots reported marijuana production only two months after it had received its vertically integrated license, according to Albuquerque Business First.
Because cannabis takes longer than two months to be cultivated, the CCD was suspicious of the report.
After inspecting the company’s premises, the CCD alleged Golden Roots reported cultivation, transportation and sales in BioTrack but wasn’t able to provide officers who inspected the site evidence that the operator was cultivating cannabis.
In April, the CCD revoked Golden Roots’ license and issued a $10,000 fine, according to the news outlet.
The company requested a hearing, which the regulator scheduled for Sept. 1.
At the hearing, the CCD testified that the company’s dried-cannabis weights in BioTrack were suspicious because they didn’t seem to accurately reflect how much weight marijuana loses as it dries.
Company employees also poorly accounted for why they were making adjustments in the system of up to 24 pounds of cannabis, making comments such as “bad” or “mistake” in the software, Albuquerque Business First reported.
Golden Roots was fined $298,972.05 – the amount regulators allege the company made selling products that weren’t properly accounted for in BioTrack.
The CCD has been cracking down on cannabis operators accused of selling products procured from out-of-state or not grown legally:
- Regulators alleged in August that Albuquerque dispensary Sawmill Sweet Leaf sold out-of-state products and didn’t have a license for extraction.
- Paradise Exotics Distro lost its license in July after regulators alleged the company sold products made in California.
Golden Roots was the first alleged rulebreaker in New Mexico to be asked to pay a large fine.
Source: https://mjbizdaily.com/new-mexico-cannabis-operator-fined-loses-license-for-alleged-biotrack-fraud/
Business
Alabama to make another attempt Dec. 1 to award medical cannabis licenses

Alabama regulators are targeting Dec. 1 to award the first batch of medical cannabis business licenses after the agency’s first two attempts were scrapped because of scoring errors and litigation.
The first licenses will be awarded to individual cultivators, delivery providers, processors, dispensaries and state testing labs, according to the Alabama Medical Cannabis Commission (AMCC).
Then, on Dec. 12, the AMCC will award licenses for vertically integrated operations, a designation set primarily for multistate operators.
Licenses are expected to be handed out 28 days after they have been awarded, so MMJ production could begin in early January, according to the Alabama Daily News.
That means MMJ products could be available for patients around early March, an AMCC spokesperson told the media outlet.
Regulators initially awarded 21 business licenses in June, only to void them after applicants alleged inconsistencies with how the applications were scored.
Then, in August, the state awarded 24 different licenses – 19 went to June recipients – only to reverse themselves again and scratch those licenses after spurned applicants filed lawsuits.
A state judge dismissed a lawsuit filed by Chicago-based MSO Verano Holdings Corp., but another lawsuit is pending.
Source: https://mjbizdaily.com/alabama-plans-to-award-medical-cannabis-licenses-dec-1/
-

 Business3 years ago
Business3 years agoPot Odor Does Not Justify Probable Cause for Vehicle Searches, Minnesota Court Affirms
-

 Business3 years ago
Business3 years agoNew Mexico cannabis operator fined, loses license for alleged BioTrack fraud
-

 Business3 years ago
Business3 years agoAlabama to make another attempt Dec. 1 to award medical cannabis licenses
-

 Business3 years ago
Business3 years agoWashington State Pays Out $9.4 Million in Refunds Relating to Drug Convictions
-

 Business3 years ago
Business3 years agoMarijuana companies suing US attorney general in federal prohibition challenge
-

 Business3 years ago
Business3 years agoLegal Marijuana Handed A Nothing Burger From NY State
-

 Business3 years ago
Business3 years agoCan Cannabis Help Seasonal Depression
-

 Blogs3 years ago
Blogs3 years agoCannabis Art Is Flourishing On Etsy













