Business
FDA Approves Nasal Spray To Reverse Fentanyl Overdoses
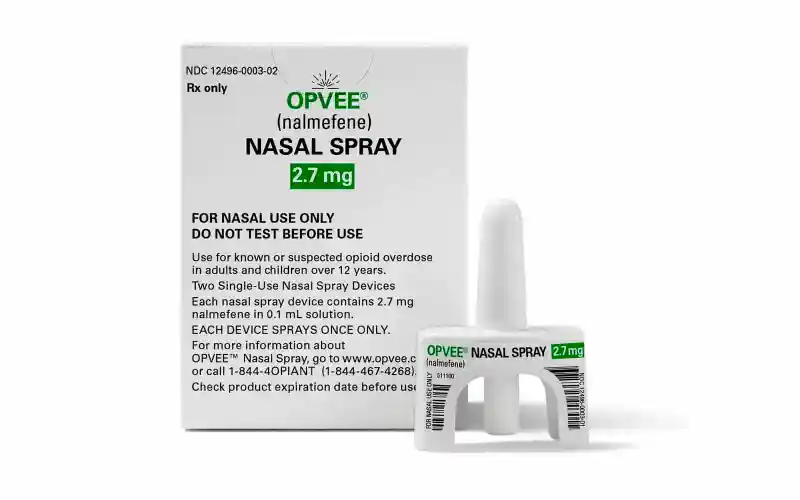
The spray, known as Opvee, has achieved similar results to Narcan in studies.
The Food and Drug Administration on Monday said that it had given regulatory approval to a nasal spray that has proven effective in reversing overdoses caused by fentanyl and other opioids.
The spray, known as Opvee, is the “the first nalmefene hydrochloride nasal spray for the emergency treatment of known or suspected opioid overdose in adults and pediatric patients 12 years of age and older,” the FDA said in the announcement, adding that it is also the “first FDA approval of nalmefene hydrochloride nasal spray for health care and community use.”
The approval is yet another step by policymakers in the United States to stem the tide of a nationwide drug crisis. Earlier this month, the U.S. Centers for Disease Control and Prevention reported that overdoses from fentanyl have spiked dramatically in recent years.
In Monday’s announcement, the FDA said that drug overdose “persists as a major public health issue in the United States, with more than 103,000 reported fatal overdoses occurring in the 12-month period ending in November 2022, primarily driven by synthetic opioids like illicit fentanyl.”
It is also part of the FDA’s “Overdose Prevention Framework,” a program launched last year “to undertake impactful, creative actions to prevent drug overdoses and reduce deaths.” Earlier this year, the FDA approved the first overdose-reversal product that can be obtained without a prescription.
“The agency continues to advance the FDA Overdose Prevention Framework and take actionable steps that encourage harm reduction by supporting the development of novel overdose reversal products,” FDA Commissioner Robert M. Califf said in Monday’s announcement.
“On the heels of the FDA’s recent approval of the first over-the-counter opioid reversal agent, the availability of nalmefene nasal spray places a new prescription opioid reversal option in the hands of communities, harm reduction groups and emergency responders.”
The opioid crisis in the United States has prompted lawmakers throughout the country to improve access to potentially life-saving drugs that can be used in the event of an overdose. The best-known is naloxone, which “has been used for decades to quickly counter overdoses of heroin, fentanyl and prescription painkillers,” according to the Associated Press.
Opvee works similarly to naloxone, the AP said, and it has “achieved similar recovery results to Narcan, the leading brand of naloxone nasal spray.”
“The approval of Opvee was supported by safety and pharmacokinetic studies, as well as a study in people who use opioids recreationally to assess how quickly the drug works. The most common adverse reactions include nasal discomfort, headache, nausea, dizziness, hot flush, vomiting, anxiety, fatigue, nasal congestion and throat irritation, pain in the nose (rhinalgia), decreased appetite, skin redness (erythema) and excessive sweating (hyperhidrosis). The use of nalmefene hydrochloride in patients who are opioid-dependent may result in opioid withdrawal characterized by the following signs and symptoms: body aches, diarrhea, fast heart rate (tachycardia), fever, runny nose, sneezing, goosebumps (piloerection), sweating, yawning, nausea or vomiting, nervousness, restlessness or irritability, shivering or trembling, abdominal cramps, weakness and increased blood pressure.”
In Minnesota, lawmakers are pushing to make Narcan available in schools.
“We simply cannot tolerate more needless loss of life. We have to act with urgency and we have to act now,” said Minnesota state Sen. Kelly Morrison, a Democrat, who is sponsoring the bill.
In its report released earlier this month, the CDC found that 69,943 people died of a fentanyl-induced overdose in 2021, which equals to a rate of 21.6 and is up considerably from 2016, when 18,499 died of an overdose from fentanyl at a rate of 5.7.
Source: https://hightimes.com/news/fda-approves-nasal-spray-to-reverse-fentanyl-overdoses/
Business
EU Pressure Builds on Google as Regulators Face Calls for Massive Fine Over Search Practices

A growing coalition of European industry groups is intensifying pressure on regulators to take decisive action against Google over allegations of unfair search practices that could reshape competition rules across the region’s digital economy.
Investigation Under Digital Markets Act Gains Momentum
The case is being examined by the European Commission under the European Union’s landmark Digital Markets Act (DMA), introduced to curb the dominance of major technology platforms and ensure fair competition.
Launched in March 2024, the investigation focuses on whether Google has been prioritising its own services in search results, potentially disadvantaging rival businesses that rely on online visibility to reach customers.
Industry Groups Demand Swift Action
Several prominent European organizations have jointly urged regulators to conclude the probe without further delay. They argue that prolonged investigations allow alleged anti-competitive practices to continue, putting European companies—especially startups—at a disadvantage.
Signatories include the European Publishers Council, the European Magazine Media Association, the European Tech Alliance, and EU Travel Tech.
In a joint statement, these groups warned that delays in enforcement are affecting innovation, profitability, and growth prospects for regional businesses competing in digital markets.
Google Denies Allegations
Google has rejected claims of bias, stating that its search algorithms are designed to deliver the most relevant and useful results to users. The company has also proposed adjustments to address regulatory concerns.
However, critics argue that these changes are insufficient and fail to address the core issue of market dominance.
Potential Billion-Euro Penalties
If found in violation of the DMA, Google could face significant financial penalties. Under EU rules, fines can reach a substantial percentage of a company’s global turnover, potentially amounting to billions of euros.
Regulators may also impose corrective measures requiring changes to business practices, which could have long-term implications for how digital platforms operate in Europe.
Wider Implications for Big Tech
The case highlights ongoing tensions between European regulators and major U.S. technology firms. In recent years, the EU has taken a more aggressive stance in enforcing competition laws, aiming to create a level playing field for local businesses.
A final ruling against Google could set a major precedent, influencing future enforcement actions and shaping the regulatory landscape for global tech companies operating within Europe.
As scrutiny intensifies, the outcome of the investigation is expected to play a critical role in defining the future of digital competition across the European Union.
AI & Technology
Amazon Faces Potential Criminal Trial in Italy Over €1.2 Billion Tax Evasion Allegations

Milan: U.S. tech giant Amazon is facing the prospect of a major legal showdown in Italy, after prosecutors in Milan formally requested a court to move forward with criminal proceedings over alleged tax evasion totaling approximately ₹12,500 crore (€1.2 billion).
The case targets Amazon’s European division along with four senior executives, marking one of the most significant tax-related investigations involving a global e-commerce platform in Europe.
Trial Push Despite Multi-Million Euro Settlement
The move comes even after Amazon reached a financial settlement with Italian tax authorities in December, agreeing to pay around ₹5,500 crore (€527 million), including interest, to resolve part of the dispute.
Typically, such settlements lead to the closure of criminal investigations. However, Milan prosecutors have opted to proceed, signaling a tougher stance on alleged corporate tax violations.
A preliminary hearing is expected in the coming months, where a judge will decide whether to formally indict the company and its executives or dismiss the case.
Allegations of VAT Evasion Through Marketplace Sellers
At the center of the investigation are claims that Amazon’s platform enabled non-European Union sellers to avoid paying value-added tax (VAT) on goods sold to Italian consumers between 2019 and 2021.
Prosecutors allege that the company’s marketplace structure allowed thousands of foreign vendors—many reportedly based in China—to operate without fully disclosing their identities or tax obligations. This, authorities argue, led to substantial VAT losses for the Italian government.
Under Italian law, online platforms facilitating sales can be held partially liable if third-party sellers fail to comply with tax requirements, a key point in the prosecution’s case.
Italian Government Named as Affected Party
In their filing, prosecutors identified Italy’s Economy Ministry as the injured party, citing significant financial damage resulting from the alleged tax evasion.
Legal experts say the outcome of the case could have wide-ranging implications across the European Union, where VAT systems are harmonized and similar compliance rules apply to digital marketplaces.
Multiple Investigations Add to Pressure
The VAT probe is just one of several legal challenges facing Amazon in Italy. The European Public Prosecutor’s Office is reportedly examining additional tax-related issues covering more recent years.
Meanwhile, Milan authorities are pursuing separate investigations into alleged customs fraud linked to imports from China and whether Amazon maintained an undeclared “permanent establishment” in Italy—potentially exposing it to higher tax liabilities.
In a separate regulatory action, Italy’s data protection authority recently ordered an Amazon unit to stop using personal data from over 1,800 employees at a warehouse near Rome.
Amazon Denies Allegations
Amazon has consistently denied wrongdoing and indicated it will strongly contest the allegations in court if the case proceeds. The company has also warned that prolonged legal uncertainty could impact investor confidence and Italy’s appeal as a destination for international business.
Broader Impact on Europe’s Digital Economy
If the case moves to trial, it could become a landmark moment for the regulation of global e-commerce platforms in Europe. Governments across the region are increasingly scrutinizing how digital marketplaces handle tax compliance, especially in cross-border transactions.
With online retail continuing to expand, regulators are under mounting pressure to ensure that multinational platforms and third-party sellers adhere to the same tax rules as traditional businesses.
Aviation
IndiGo Crisis Exposes Risks of Monopoly: What If Telecom or E-commerce Collapses Next?

Airports across India witnessed scenes of distress and confusion as thousands of passengers were stranded due to IndiGo’s massive flight disruptions. Families with medical emergencies, funerals, and personal crises were left helpless as the airline cancelled hundreds of flights without adequate communication or support.
Passengers described desperate situations — a mother pleading for sanitary pads for her daughter, a woman unable to transport her husband’s coffin, and others stranded while trying to reach family funerals or hospitals. “It was like a lockdown at the airport,” one passenger said, describing the panic that unfolded as IndiGo’s mismanagement crippled operations nationwide.
Root Cause: IndiGo’s Market Monopoly
The turmoil, industry experts argue, stems from IndiGo’s monopolistic control over India’s domestic aviation market. The airline operates nearly 2,100 flights daily and holds around 60% market share — meaning every second plane flying within India belongs to IndiGo.
This dominance has given the company unparalleled influence. When IndiGo falters, the entire aviation system suffers. Passengers are left with few alternatives, as other airlines lack capacity to absorb stranded travellers. The result: skyrocketing ticket prices, chaos at terminals, and total dependence on a single private operator.
Aviation pioneer Captain G.R. Gopinath, founder of Air Deccan, criticised the government’s inaction, noting that on some routes, IndiGo’s economy fares surged to ₹1 lakh. He compared the situation to a hostage crisis, writing that the airline “held the system ransom” and forced regulators to defer new safety rules meant to protect pilots and passengers.
Government Intervention and Regulatory Weakness
The crisis erupted after IndiGo failed to comply with the Flight Duty Time Limitations (FDTL) — rules introduced by the DGCA in January 2024 requiring adequate rest for pilots. Despite having nearly two years to adapt, IndiGo blamed the rule for operational disruptions, citing a shortage of pilots.
Under mounting public pressure, the government stepped in, temporarily relaxing FDTL norms and capping airfare hikes. Officials claimed the move was to protect passengers, but analysts say it exposed the state’s vulnerability to corporate monopolies. “The government had no option but to yield,” said one aviation policy expert, pointing out that ignoring safety regulations for short-term relief could have long-term consequences.
The crisis also rekindled memories of the June 2025 Air India crash near London, which claimed over 240 lives. Experts warn that compromising pilot rest and safety standards to maintain flight schedules could risk another tragedy.
If Telecom Giants Fail: A National Paralysis
The article raises a troubling question — what if a similar crisis struck the telecom sector, where Jio and Airtel together control nearly 80% of subscribers and serve over 780 million users?
If both networks failed simultaneously, the repercussions would be catastrophic. Internet shutdowns would halt UPI transactions, online banking, OTP verifications, video calls, OTT streaming, and emergency communications. Critical services such as airports, hospitals, stock exchanges, and small businesses — many of which rely on WhatsApp and digital payments — would come to a standstill.
In essence, a telecom breakdown could paralyse India’s digital economy, exposing the nation’s dependence on a duopoly.
E-commerce Monopoly: Another Fragile Ecosystem
The same risk looms over the e-commerce sector, where Amazon and Flipkart dominate nearly 80% of the market. A disruption similar to IndiGo’s could cripple daily life — halting delivery of groceries, medicines, and essential goods, freezing refunds and customer support, and leaving small sellers without platforms to trade.
Local retailers, freed from competition, might exploit shortages by inflating prices. Such a scenario underscores the perils of market centralisation in sectors critical to everyday living.
A Wake-Up Call for Regulators
The IndiGo crisis, analysts say, is a warning shot for policymakers and regulators. A single company’s operational failure exposed systemic weaknesses in India’s infrastructure and consumer protection mechanisms.
As the aviation regulator DGCA investigates and IndiGo works to restore normalcy, the broader lesson remains clear: unchecked monopoly power in any essential service — whether air travel, telecom, or e-commerce — poses a direct threat to economic stability and citizen welfare.
Without stronger competition laws, redundancy frameworks, and regulatory oversight, India risks repeating this crisis across multiple sectors — each time with millions of citizens paying the price.
-

 Business3 years ago
Business3 years agoPot Odor Does Not Justify Probable Cause for Vehicle Searches, Minnesota Court Affirms
-

 Business3 years ago
Business3 years agoNew Mexico cannabis operator fined, loses license for alleged BioTrack fraud
-

 Business3 years ago
Business3 years agoAlabama to make another attempt Dec. 1 to award medical cannabis licenses
-

 Business3 years ago
Business3 years agoWashington State Pays Out $9.4 Million in Refunds Relating to Drug Convictions
-

 Business3 years ago
Business3 years agoMarijuana companies suing US attorney general in federal prohibition challenge
-

 Business3 years ago
Business3 years agoLegal Marijuana Handed A Nothing Burger From NY State
-

 Business3 years ago
Business3 years agoCan Cannabis Help Seasonal Depression
-

 Blogs3 years ago
Blogs3 years agoCannabis Art Is Flourishing On Etsy













