Government
NYC’s Overdose Prevention Centers Prove Effective
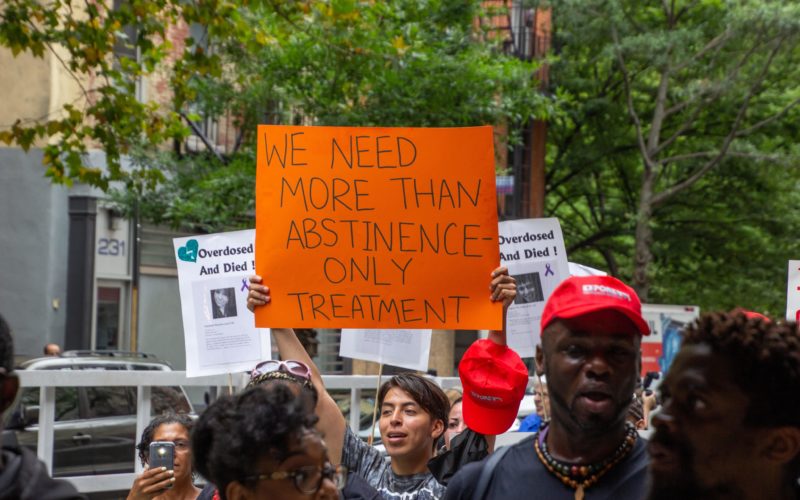
New research shows the first publicly recognized centers in the nation reduced overdoses.
A new study published this month has found that New York City’s historic safe consumption centers have helped reduce overdoses.
The study, conducted by researchers affiliated with the NYC Department of Health (which oversees the sites), covered the two months of the program across two different consumption sites.
Last November, then-New York Mayor Bill de Blasio announced “that the first publicly recognized Overdose Prevention Center (OPC) services in the nation have commenced in New York City.”
OPCs, the city explained in the announcement, “are safe places where people who use drugs can receive medical care and be connected to treatment and social services.”
NYC officials touted their effectiveness, saying in the press release at the time that such services are “proven to prevent overdose deaths, and are in use in jurisdictions around the world,” and that there “has never been an overdose death in any OPC.”
A study from the city’s Department of Health found that “OPCs in New York City would save up to 130 lives a year.”
“New York City has led the nation’s battle against COVID-19, and the fight to keep our community safe doesn’t stop there. After exhaustive study, we know the right path forward to protect the most vulnerable people in our city. And we will not hesitate to take it,” de Blasio said in the announcement. “Overdose Prevention Centers are a safe and effective way to address the opioid crisis. I’m proud to show cities in this country that after decades of failure, a smarter approach is possible.”
The study published this month may be seen as vindication for the advocates of the program.
“During the first 2 months of OPC operation, trained staff responded 125 times to mitigate overdose risk. In response to opioid-involved symptoms of overdose, naloxone was administered 19 times and oxygen 35 times, while respiration or blood oxygen levels were monitored 26 times,” the authors wrote. “In response to stimulant-involved symptoms of overdose (also known as overamping), staff intervened 45 times to provide hydration, cooling, and de-escalation as needed. Emergency medical services responded 5 times, and participants were transported to emergency departments 3 times. No fatal overdoses occurred in OPCs or among individuals transported to hospitals.”
“This quality improvement study found that during the first 2 months of operations, services at 2 OPCs in NYC were heavily used, with early data suggesting that supervised consumption in these settings was associated with decreased overdose risk,” they added. “Data also suggested that OPCs were associated with decreased prevalence of public drug use.”
The authors did, however, caution that the findings are “limited by the short study period and lack of a comparison group with individuals not participating in OPC services,” and that additional “evaluation may explore whether OPC services are associated with improved overall health outcomes for participants, as well as neighborhood-level outcomes, including public drug use, improperly discarded syringes, and drug-related crime.”
But the study provides hope to those who are desperate to mitigate an overdose crisis that has become a national epidemic in the United States.
In the announcement of the OPC services last November, NYC officials said that “over 2,000 individuals died of a drug overdose in New York City [in 2020], the highest number since reporting began in 2000,” and that the “Centers for Disease Control projects that across the United States, more than 90,000 individuals died of a drug overdose during 2020, the worst year on record.”
Citing self-reported data, the authors of the new study said that “the drug most commonly used across 2 sites was heroin or fentanyl (73.7%) and the most frequent route of drug administration at the OPC was injection (65.0%).”
Source: https://hightimes.com/news/nycs-overdose-prevention-centers-prove-effective/
Business
New Mexico cannabis operator fined, loses license for alleged BioTrack fraud

New Mexico regulators fined a cannabis operator nearly $300,000 and revoked its license after the company allegedly created fake reports in the state’s traceability software.
The New Mexico Cannabis Control Division (CCD) accused marijuana manufacturer and retailer Golden Roots of 11 violations, according to Albuquerque Business First.
Golden Roots operates the The Cannabis Revolution Dispensary.
The majority of the violations are related to the Albuquerque company’s improper use of BioTrack, which has been New Mexico’s track-and-trace vendor since 2015.
The CCD alleges Golden Roots reported marijuana production only two months after it had received its vertically integrated license, according to Albuquerque Business First.
Because cannabis takes longer than two months to be cultivated, the CCD was suspicious of the report.
After inspecting the company’s premises, the CCD alleged Golden Roots reported cultivation, transportation and sales in BioTrack but wasn’t able to provide officers who inspected the site evidence that the operator was cultivating cannabis.
In April, the CCD revoked Golden Roots’ license and issued a $10,000 fine, according to the news outlet.
The company requested a hearing, which the regulator scheduled for Sept. 1.
At the hearing, the CCD testified that the company’s dried-cannabis weights in BioTrack were suspicious because they didn’t seem to accurately reflect how much weight marijuana loses as it dries.
Company employees also poorly accounted for why they were making adjustments in the system of up to 24 pounds of cannabis, making comments such as “bad” or “mistake” in the software, Albuquerque Business First reported.
Golden Roots was fined $298,972.05 – the amount regulators allege the company made selling products that weren’t properly accounted for in BioTrack.
The CCD has been cracking down on cannabis operators accused of selling products procured from out-of-state or not grown legally:
- Regulators alleged in August that Albuquerque dispensary Sawmill Sweet Leaf sold out-of-state products and didn’t have a license for extraction.
- Paradise Exotics Distro lost its license in July after regulators alleged the company sold products made in California.
Golden Roots was the first alleged rulebreaker in New Mexico to be asked to pay a large fine.
Source: https://mjbizdaily.com/new-mexico-cannabis-operator-fined-loses-license-for-alleged-biotrack-fraud/
Business
Marijuana companies suing US attorney general in federal prohibition challenge

Four marijuana companies, including a multistate operator, have filed a lawsuit against U.S. Attorney General Merrick Garland in which they allege the federal MJ prohibition under the Controlled Substances Act is no longer constitutional.
According to the complaint, filed Thursday in U.S. District Court in Massachusetts, retailer Canna Provisions, Treevit delivery service CEO Gyasi Sellers, cultivator Wiseacre Farm and MSO Verano Holdings Corp. are all harmed by “the federal government’s unconstitutional ban on cultivating, manufacturing, distributing, or possessing intrastate marijuana.”
Verano is headquartered in Chicago but has operations in Massachusetts; the other three operators are based in Massachusetts.
The lawsuit seeks a ruling that the “Controlled Substances Act is unconstitutional as applied to the intrastate cultivation, manufacture, possession, and distribution of marijuana pursuant to state law.”
The companies want the case to go before the U.S. Supreme Court.
They hired prominent law firm Boies Schiller Flexner to represent them.
The New York-based firm’s principal is David Boies, whose former clients include Microsoft, former presidential candidate Al Gore and Elizabeth Holmes’ disgraced startup Theranos.
Similar challenges to the federal Controlled Substances Act (CSA) have failed.
One such challenge led to a landmark Supreme Court decision in 2005.
In Gonzalez vs. Raich, the highest court in the United States ruled in a 6-3 decision that the commerce clause of the U.S. Constitution gave Congress the power to outlaw marijuana federally, even though state laws allow the cultivation and sale of cannabis.
In the 18 years since that ruling, 23 states and the District of Columbia have legalized adult-use marijuana and the federal government has allowed a multibillion-dollar cannabis industry to thrive.
Since both Congress and the U.S. Department of Justice, currently headed by Garland, have declined to intervene in state-licensed marijuana markets, the key facts that led to the Supreme Court’s 2005 ruling “no longer apply,” Boies said in a statement Thursday.
“The Supreme Court has since made clear that the federal government lacks the authority to regulate purely intrastate commerce,” Boies said.
“Moreover, the facts on which those precedents are based are no longer true.”
Verano President Darren Weiss said in a statement the company is “prepared to bring this case all the way to the Supreme Court in order to align federal law with how Congress has acted for years.”
While the Biden administration’s push to reschedule marijuana would help solve marijuana operators’ federal tax woes, neither rescheduling nor modest Congressional reforms such as the SAFER Banking Act “solve the fundamental issue,” Weiss added.
“The application of the CSA to lawful state-run cannabis business is an unconstitutional overreach on state sovereignty that has led to decades of harm, failed businesses, lost jobs, and unsafe working conditions.”
Business
Alabama to make another attempt Dec. 1 to award medical cannabis licenses

Alabama regulators are targeting Dec. 1 to award the first batch of medical cannabis business licenses after the agency’s first two attempts were scrapped because of scoring errors and litigation.
The first licenses will be awarded to individual cultivators, delivery providers, processors, dispensaries and state testing labs, according to the Alabama Medical Cannabis Commission (AMCC).
Then, on Dec. 12, the AMCC will award licenses for vertically integrated operations, a designation set primarily for multistate operators.
Licenses are expected to be handed out 28 days after they have been awarded, so MMJ production could begin in early January, according to the Alabama Daily News.
That means MMJ products could be available for patients around early March, an AMCC spokesperson told the media outlet.
Regulators initially awarded 21 business licenses in June, only to void them after applicants alleged inconsistencies with how the applications were scored.
Then, in August, the state awarded 24 different licenses – 19 went to June recipients – only to reverse themselves again and scratch those licenses after spurned applicants filed lawsuits.
A state judge dismissed a lawsuit filed by Chicago-based MSO Verano Holdings Corp., but another lawsuit is pending.
Source: https://mjbizdaily.com/alabama-plans-to-award-medical-cannabis-licenses-dec-1/
-

 Business2 years ago
Business2 years agoPot Odor Does Not Justify Probable Cause for Vehicle Searches, Minnesota Court Affirms
-

 Business2 years ago
Business2 years agoNew Mexico cannabis operator fined, loses license for alleged BioTrack fraud
-

 Business2 years ago
Business2 years agoAlabama to make another attempt Dec. 1 to award medical cannabis licenses
-

 Business2 years ago
Business2 years agoWashington State Pays Out $9.4 Million in Refunds Relating to Drug Convictions
-

 Business2 years ago
Business2 years agoMarijuana companies suing US attorney general in federal prohibition challenge
-

 Business2 years ago
Business2 years agoLegal Marijuana Handed A Nothing Burger From NY State
-

 Business2 years ago
Business2 years agoCan Cannabis Help Seasonal Depression
-

 Blogs2 years ago
Blogs2 years agoCannabis Art Is Flourishing On Etsy











